By Kenneth Richard on 9. May 2019
Another new paper published in Paleoceanography and Paleoclimatology casts further doubt on the paradigm that says CO2 has historically been a temperature driver.
Evidence from the tropical Atlantic indicates today’s regional temperatures (15.5°C) are 7.5°C colder than a peak temperatures (23°C) between 15,000 to 10,000 years ago, when CO2 hovered around 220 ppm.

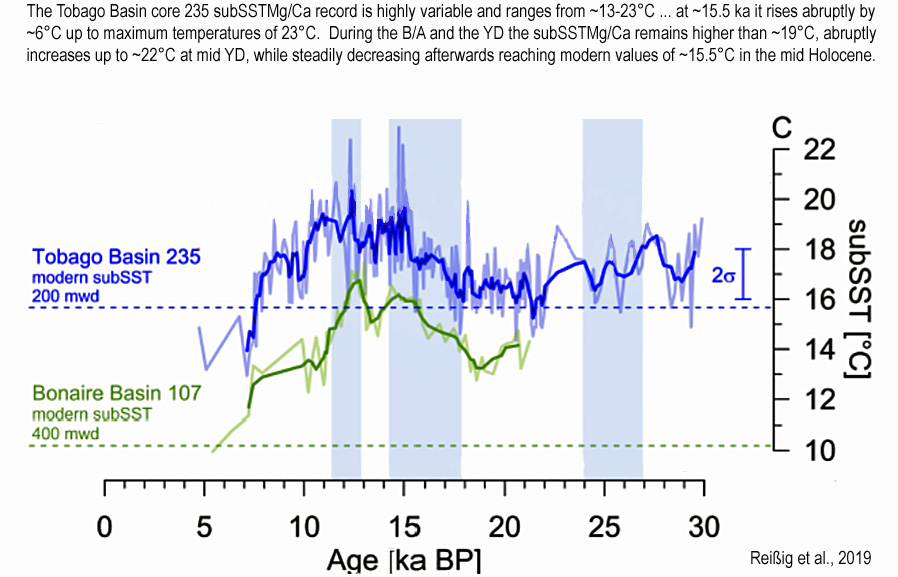
“[T]he Tobago Basin core 235 subSSTMg/Ca record is highly variable and ranges from ~13-23°C, which is approximately three times as much as at Beata Ridge.. In Tobago Basin, the subSSTMg/Ca decrease by ~2°C from 30 ka BP (18°C) to the onset of HS1 (16°C). Within HS1, the subSSTMg/Ca increase continuously by 2°C, while at ~15.5 ka it rises abruptly by ~6°C up to maximum temperatures of 23°C. The abrupt subSST rise is delayed too the reconstructed SST rise at the beginning of HS1 by Bahr et al. (2018) (Fig. S7). Subsequently, subSSTMg/Ca scatters around 20°C until the beginning of the Bølling-Allerød (B/A). During the B/A and the YD the subSSTMg/Ca remains higher than ~19°C, abruptly increases up to ~22°C at mid YD, while steadily decreasing afterwards reaching modern values of ~15.5°C in the mid Holocene. Lowest subSSTMg/Ca of ~13°C are observed after ~7 ka BP. On average, the LGM subSSTMg/Ca are warmer by ~2.5°C than during the Holocene.”
“[T]he subsurface temperature variability is a robust climate signal in the tropical W Atlantic. Both records show an increase of ~5°C in subSSTMg/Ca from the LGM to the early YD and a subSSTMg/Ca decrease by ~7-8°C during the Holocene suggesting that both sediment cores are influenced by the same oceanographic changes. Notably, the mid Holocene subSSTMg/Ca in Tobago and Bonaire Basins remain cooler by ~1.5°C and ~3°C, respectively, than during the LGM.”
“At Tobago Basin and Bonaire Basin, the deglaciation is characterized by abrupt rises in subSSTMg/Ca by ~5.5°C at the end of HS1 and by ~6°C at the middle of the YD to peak values of up to ~23°C and ~22°C, respectively, accompanied by changes towards saline conditions (mean δ18Osw-ivf of ~2.25‰ and ~2‰, respectively (Fig. 3). These highly variable changes occur within less than 400 years.”
“In contrast to modern conditions Tobago Basin core 235 was influenced by a warm water mass between 30-10 ka BP, indicated by elevated subSSTMg/Ca (~2.5°C warmer than the modern conditions).”
Posted in Paleo-climatology |







I missed the article downthread where it was pointed that seawater temperature affects the partial pressure of CO2. Now this demonstrating the variability of SUBSSTMg/Ca. I am convinced that increasing CO2 levels cannot with any confidence be tied to human activity without first quantifying the natural sources.
The effect of seawater temperature is largely quantified. Without any human made increase of the CO2 concentration this would certainly cause the concentration to increase a bit. But with the additional human CO2 the partial pressure in the atmosphere is higher than in the oceans and thus the oceans become a net absorber of CO2 from the atmosphere, despite warming up considerably.
The science behind this is solid.
Regarding the post itself:
This sounds like you, Kenneth, are implying that CO2 has no effect nowbecause it didn’t cause a warming in the past. Is that what you are trying to say?
Well, good news then … it appears like the CO2 concentration is also not causing the daytime/nighttime temperature difference. Another nail in CO2’s coffin, right? 😉
I wonder if you really read what seems to have caused this subsurface warming in the region?
So it’s a shift of heat towards a certain region. How on Earth does that show that CO2 doesn’t cause the modern warming?
CO2 has an effect. I just don’t know what it is since we have no observational evidence that CO2 heats up water when increased. It could be 0.000000000001 K per 100 ppm or 0.0000000000000000000000001 K per 100 ppm. Either way, there’s an effect. And there’s no way to know which value is closer because the believers haven’t bothered to actually test their assumption. That way it’s unfalsifiable. There’s no way to disprove that which hasn’t been observed. So no matter what, you’re right.
Gravity has an effect. I just don’t know what it is since we have no observational evidence that gravity can actually influence the path of a probe in another solar system.
That’s how this “argument” sounds like.
It’s a range with probability derived from countless actual tests.
It’s not.
It has been observed, evidence strongly points to this, yet there exists a niche which strongly believes in some kind of conspiracy or mass dellusion regarding this topic because “they know better” and feel like there is actual evidence supporting a doubt in what science in general found to be what is happening.
What is really no there is a natural variability that is causing the current climate change. You’ll probably come to terms with this in a few decades and ask yourself how you could ever see it the way you do now. Want to bet?
Silly comment, Seb. But it doesn’t surprise any of us here that you’d think scientists have determined CO2’s effect to be some sort of universal constant like gravity. Sometimes I think you’re a full-blown schmuck.
Silly comment is all yours, Pierre. Since when is gravity a universal constant?
In the core SebH says that Oceans become a “net absorber… despite warming “and he quotes the science is settled.
This would mean there is a calculation available to back up this claim.
If he could show us a paper that actually backs him up, would be helpful.
The casual observer would need this, because it is known that the oceans outgas CO2 when getting warmer so they are first of all a source. How would a change of a couple of ppm on atmospheric CO2 be able to overcome this outgassing or does it even, is the question.
If the science was settled this could be put down in the books and should not be argued about.
SebH, mind me asking what your sources for your claim are?
Also please be so kind and explain if you are aware that the cooler oceans do most of the absorbing and are responsible for the CO2 uptake, while the warmer oceans do the outgassing.
Please explain how a warmer ocean effects corals, by outgassing of CO2, which in my books changes the pH to the contrary of what you would claim.
Also explain how it comes that we rely on Mauna Loa CO2 measurements, when clearly this location is where the oceans are a source of CO2.
If the science is settled, then show us the evidence and the papers.
This is just basic physics, John Brown. A textbook about partial pressure should suffice, I doubt that anyone recently wrote a paper about that.
When the system is in balance the outgassing in the tropics equal out with the absorption towards the poles. We seem to agree that temperature somewhat controls the outgassing/absorption part of the equation, right? Now, partial pressure is also part of the equation, John. If the atmosphere would suddenly consist of 100% CO2 you surely would agree that the oceans would absorb the CO2 until the partial pressure in both reservoirs equals out. This also happens at much lower concentrations.
Look up Henry’s law if you don’t “believe me”. The Wikipedia article on it is quite helpful: https://en.wikipedia.org/wiki/Henry%27s_law (pay attention to the section about temperature dependence).
It actually is “in the books”. I don’t know why skeptics try to imagine that ocean outgassing due to warming would be the driver of the CO2 concentration increase just because the seasonal CO2 concentration changes correlate with ocean surface temperature changes.
We are all aware. Again, the net result of the partial pressure difference is absorption even though the oceans warm up. That doesn’t mean that the oceans are suddenly absorbing CO2 from the atmosphere in warm regions. Again, study Henry’s law. Basic physics.
Do you honestly believe that this is the only place CO2 gets measured? If I were a skeptic in the sense you guys are, I’d be more worried about the volcanoes in that area than ocean outgassing.
In case the mention of Henry’s law is not enough, here is one paper about the ocean-atmosphere CO2 fluxes:
https://agupubs.onlinelibrary.wiley.com/doi/full/10.1002/2016GB005400
Can you believe what the author wrote in that paper or will you argue that it’s nonsense since it’s just a model/simulation based on basic laws of physics?
I said it before and I say it again. Henrys law is massively incompetent to describe the Carbon flux into the oceans.
The main problem is that CO2 when introduced into water dissociates as part of a chemical reaction. This limits the usefulness when trying to describe the uptake of carbon from the oceans, which also has been described in the paper that you suggested.
From the summary:
“Drivers of the air‐sea CO2 flux are (1) The effect of heat and freshwater fluxes on the saturated ocean carbon concentration, which depends on surface temperature, salinity, and alkalinity at steady state atmospheric pCO2; (2) the direct effect of freshwater dilution on surface CT concentrations; and (3) carbon uptake and export by biological activity (as both soft tissue and carbonate) through nutrient utilization, and atmosphere‐ocean disequilibrium, which is influenced by upwelling and entrainment of remineralized carbon‐ and nutrient‐rich waters from the ocean interior as well as rapid subduction of surface waters before reaching full equilibrium.”
There we see that the main contributors are temperature and biological life. As a side the uptake through fresh water is stipulated which would also include uptake through the solution process as described in Henrys law.
Based on this paper your sentence:
“I don’t know why skeptics try to imagine that ocean outgassing due to warming would be the driver of the CO2 concentration increase just because the seasonal CO2 concentration changes correlate with ocean surface temperature changes.”
makes no sense. Because the temperature effect is what is described in the paper you wanted me to read. Outgassing due to warming makes entirely sense.
Did you read and understand the paper? Did you also see the reference to the “simulation” part in it?
In the real world Mauna Loa is in an area where ocean temperature are said to be increasing. I think we have all the evidence to assume that the measurements there are affected by it.
As you would say, all based on the laws of physics and now also based on the paper that you referenced.
Oh dear … this is too hard to reply to from a smartphone, but way to go almost completely ignoring partial pressure differences. And of course Mauna Loa is affected … It’s a CO2 measuring station (amongst other things). Every single station is affected by outgassing AND absorption of the oceans. That’s why we see seasonal variations (biosphere plays a role as well in that) and not at all the amount of increase that would be expected from the amount of CO2 we emit into the atmosphere.
The tiny increase in outgassing and less absorption due to increasing temperatures is completely overwhelmed by the partial pressure difference. Again, this doesn’t mean that the ocean is suddenly absorbing CO2 in warm regions …
“Did you read and understand the paper?”
Did you? The very first equation IS what determines the CO2 exchange. It’s about the partial pressure difference multiplied with an efficiency factor. How can one overlook that so easily?
[…] K. Richard, May 9, 2019 in […]
It is the 10th day of May. 32 years ago the lilacs were in full bloom and the temps outside were much warmer then than now.
The lilacs aren’t going to bloom for another 5 to 10 days now. Leaf development is barely beginning here.
Translation: It’s one of the coldest spring thaws ever. Yesterday’s low morning temp was 23 degrees F. The record low for the date was recorded in 1945 at 20 degrees F.
It is not warm weather at all. The potato planting has been delayed a week or more. If you plant potatoes too early, the crop could freeze, then no potatoes. Crop failure is not good. Too cold to plant potatoes, that’s the verdict.
The sea ice extent is influencing the current weather patterns, there is a lot of sea ice at the North Pole in the here and now. the fact of the matter.
You are only fooling yourself if you think the globe is warming because humans and their burning of fossil fuels is to blame, it is not happening, at all.
The oceans should have been outgassing massive amounts of co²
oops. I was evidently under the mistaken impression that before recently, the co2 level had remained at about 280 ppmv for hundreds of thousands (if not millions) of years.
Now the story is 220 ppmv, basically during our current interglacial period, and at about the time our ice age ended.
What’s going on?
Yes Denis, that’s how it was. Nature was in perfect balance and harmony for millions of years, happy and contented with the most optimal ever level of co2. Then, evil man started up those nasty machines, and tipped innocent Nature onto a runaway course towards climate destruction. You are a Man, Denis, so you are guilty of this heinous crime. /sarc
Henry Huse @HuseEnergy One thing that is never mentioned is where atmospheric CO2 levels are measured. All of the data is from observations in the lower troposphere. Upper elevation greenhouse gas pollution has a substantial different effect on global warming. Jet aircraft pump over 800 million tons of CO2 per year in the upper atmosphere where it can remain up to 100 years. NOX adds to the effect. My hypothesis is that the resulting greenhouse gas layer reduces night time radiation, thus creating a thermodynamic imbalance. It is essential to measure the composition of the upper troposphere, which hasn’t been done.
How do radiative active gasses that are introduced into upper layers of the atmosphere reduce night time radiation, when this layer is responsible for cooling of that same atmosphere through radiation?
These additional gasses would increase radiative heat loss create a cooling effect.
Those gases don’t just radiate in the direction of space, John. More of them in any height move up the effective emission height.
And yeah, CO2 concentrations have been measured at height:
https://www.atmos-chem-phys.net/17/3861/2017/acp-17-3861-2017.pdf
Funny Seb,
it still stands to reason that more CO2 molecules will be able to radiate more energy away.
It does not matter that they radiate into the direction of the surface. They do radiate also into space.
Maybe try an analogy here, John. Will adding a second pair of sunglasses allow more sunlight to reach your eyes or less?
Or: adding more blankets to cover you increases the surface area, but will it could more heat loss to the environment as well because of that?
Or in terms of CO2 molecules … when you add more of them, they don’t just magically radiate more. How could they? Even with 100% CO2 the amount that gets radiated away will still equal the amount of energy that comes in. What changes is the surface temperature and the effective emissions height.
My hypothesis on the effect of greenhouse gases in the upper atmosphere requires verification by actually measuring the concentration, which I don’t think has been done. It is notable that summer nights in the U.S. increased twice the rate of days, with overnight lows increasing 1.4 degrees F as compared to daytime high temperatures increasing 0.7 F. This would a reduction of night time radiation. This is discussed in my blog, Hot Nights, energyconservationtech.blogspot.com.
Check out the link that SebH send in his post above.
“And yeah, CO2 concentrations have been measured at height:
https://www.atmos-chem-phys.net/17/3861/2017/acp-17-3861-2017.pdf“
[…] https://notrickszone.com/2019/05/09/new-study-the-tropical-atlantic-was-7-5c-warmer-than-now-while-c… […]
[…] https://notrickszone.com/2019/05/09/new-study-the-tropical-atlantic-was-7-5c-warmer-than-now-while-c… […]
Henry Huse The study cited by John Brown relates primarily to global CO2 distribution and provides measurements in ppmv (volume). My calculations, based on mass, indicated that the 30,000 to 38,000 altitude aircraft flight zone has about 13 % of the atmosphere’s volume and 2% of it’s mass. Adding over 800 million tons per year from aircraft works out to about 8 ppm which would be added to the greenhouse layer. The article further supports my contention that CO2 and NOX in the upper atmosphere is basically inert and can remain in orbit up to 100 years. Ref: https// energyconservationtech.blogspot.com