A new study shows ocean pH (“acidification”) levels naturally vary seasonally and decadally at rates and magnitudes far exceeding those attributed to anthropogenic activity.
In recent decades the oceans’ average pH level has fallen to 8.1 according to NOAA. This pH value is said to be about one-tenth of a unit lower than it was before modern industrialization (8.2).
Because the oceans are less alkaline than they were at one point in time, the directionality of these pH changes is referred to as an ocean acidification process. Of course, humans are said to be responsible for this. We are allegedly acidifying the oceans, or facilitating the 8.2 to 8.1 decline over the course of the last few centuries, because we humans have caused atmospheric CO2 levels to rise from 280 ppm to 415 ppm since 1750. That’s the allegation, anyway.
But these assumptions seem to be challenged by the results shown in studies authored by the very scientists promoting the human-caused ocean acidification narrative.
For example, a new study shows pH levels were lower in the 1730s and 1930s-’40s than in 2000, and that the highest pH levels (i.e., less acidic) of the last 300 years occurred in the 1960s to 1980s. Anthropogenic CO2 emissions were negligible to non-existent in the 1730s, and by the 1930s they were only hovering around 1 GtC/year. In the late 20th century, human emissions rates reached about 5 or 6 GtC/year, and yet it was these decades that had the highest pH levels since the early 18th century.
Further, seawater pH in the South China sea varies seasonally, or from about 8.1 in winter to 7.6 in summer. So, within a span of months, the ocean pH varies far more than it allegedly has due to human activity over centuries.
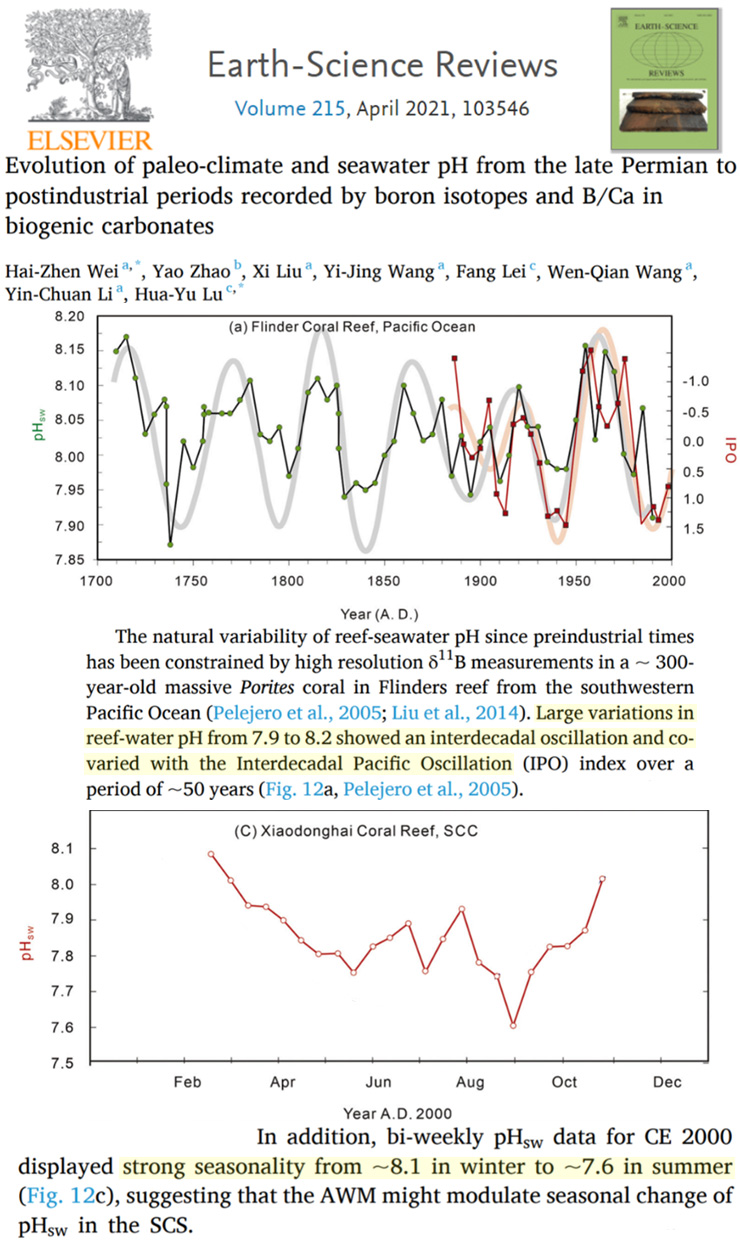
Image Source: Wei et al., 2021
A few other graphs from the Wei et al. (2021) paper show just how ordinary and unexceptional the modern atmospheric CO2 and ocean pH levels are relative to the past.
In the main graph from the below image, notice how minuscule a 400 ppm atmospheric CO2 level and 8.2 to 8.1 pH change (circles on the top left and right) look relative to the changes over the last 25 million years (Ma).
Then notice the wild variations in CO2 reaching 2500 to 3500 ppm between 40 to 60 Ma, and the pH values dipping to 7.4 or 7.5 during this same era. The top right graph shows pH levels dipping to 7.2 and atmospheric CO2 fluctuating by ±1000 ppm from about 182 to 185 Ma.
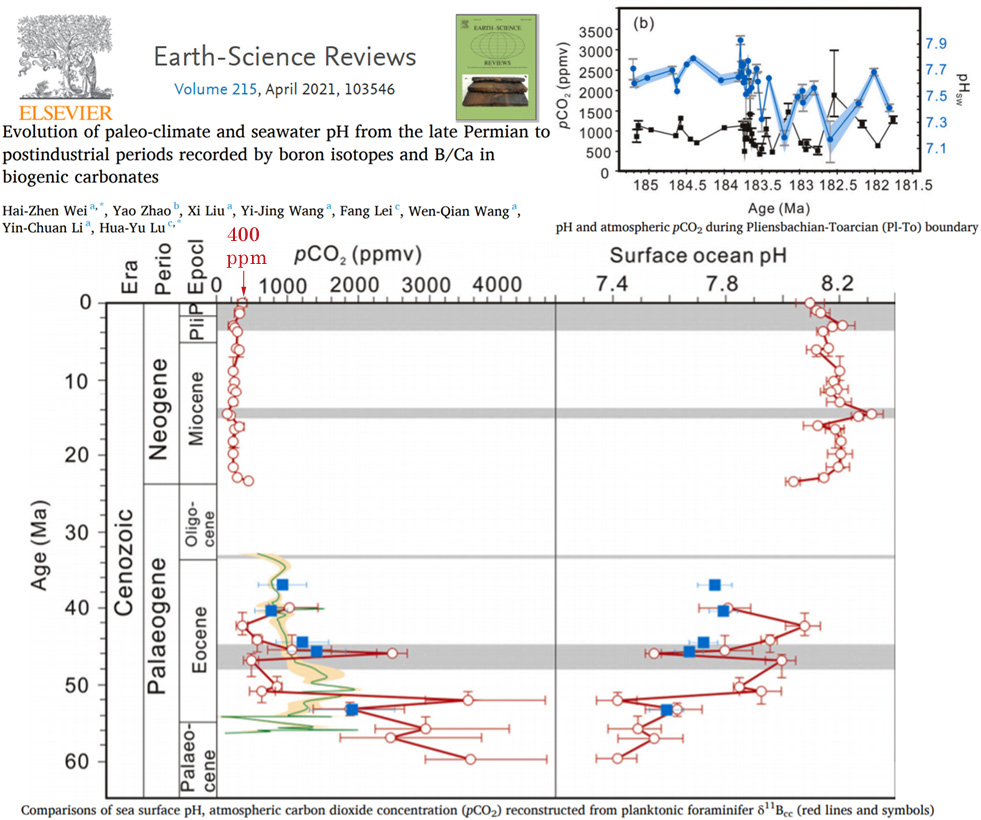
Image Source: Wei et al., 2021
Given today’s alarmism about ocean acidification driven by high atmospheric CO2 levels, these images beg the question:
How did marine animals manage to survive in such “acidified” waters (7.2 to 7.5) if it’s believed the modern biosphere is threatened by a pH value of 8.1?





Marine animals didn’t survive the 1730 acidity. They were all dissolved, and if we go on producing CO2 they’ll all be dissolved again.
[…] Related: Oceans Were More ‘Acidic’ In The 1730s…Today’s CO2 Levels Are Geologically Trifling […]
Animals that were evolved 20-40 million years ago should be able to handle reduced alkalinity in sea water (oysters, scallops); animals that have evolved in the last 2 million years (mussels) may not. When water first flowed on earth, its pH was less than 7. With increased salinity of the oceans over the millions of years since then, pH has increased with the increased salinity. As Prof Sam Carey said to me, the problem is we only live three score years and ten or thereabouts, but geological timescales are far greater.
“When water first flowed on earth, its pH was less than 7”
Common, everyday rainwater is acidic: 5.6 pH.
Rivers are sub 7 pH.
The oceans fluctuate between 6.8 and 8.1 pH within hours on a daily basis.
There is nothing unusual occurring today that hasn’t happened before.
CO2 is a trace gas in the atmosphere, which itself has a mass that’s just a tiny fraction of the ocean’s mass. The notion that changes in the concentration of atmospheric CO2 can change the acidity / alkalinity of the ocean is mythical thinking, or a lie, depending on who’s telling it.
BTW, climate alarmists also claim that the ocean is warming. If it did, it would absorb less CO2 from the atmosphere, while exhaling more CO2 into the atmosphere. In other words “global warming” and “ocean acidification” are two incompatible lies.
According to graphs furnished by NASA, the sea and the air at sea level are increasing in temperature. This invites a thought experiment that casts doubt on the possibility that more heat causes more violent storms. Imagine that weather is a heat engine with the sea as the cold side and the air being the hot side of a heat engine that has efficiency equal to (1-(Tcold/Thot)). If you add one degree Kelvin to both the numerator of the hot side and the denominator of the hot side, the efficiency goes down. As for Ph. The classic answer for this is that the sea is buffered and resists changes in Ph. The older the oceans, the more salt it acquires.
Please consider under water volcanoes. Remember that atolls form seem to
form around extinct volcanic vents. There are chains of volcanoes under the Pacific.
If atmospheric CO2 in contact ocean water can change it’s PH, how come we don’t see PH changes in fresh water lakes? Also CO2 can be slightly acidic or slightly basic, it depends on how it feels.
The volcanic vents must still be active as fish use up oxygen that must be replaced. They cannot survive a disruption.
When will all visit or revisit the actual missing 2 million ocean pH measurements? When you finally recognize that this is unacceptable, you’ll appreciate how the optics of indifference are not good. Let’s restore those measurements to the main scientific streams asap.
If any don’t understand they are free to study the topic at my site for starters such as the essay on the stories of O.
Michael Wallace